코로나바이러스 (SARS-Severe acute respiratory syndrome -CoV-2)감염병, COVID-19
1. 최근 정보
코로나바이러스 (SARS-Severe acute respiratory syndrome -CoV-2) 감염병, COVID-19
코로나 바이러스 영상
소스: 미 CDC
인간 코로나바이러스 (human coronaviruses (HCoVs)에는 7종이 있다. |
1. HCoV-229E,2. HCoV-OC43,3. HCoV-NL63,4. HCoV-HKU1,5. severe acute respiratory syndrome coronavirus (SARS-CoV),6. Middle East respiratory syndrome coronavirus (MERS-CoV)7. Novel coronavirus (2019-nCoV, a.k.a. SARS-CoV-2). |
|
SARS-CoV-2와 COVD-19는 약자이다. |
· SARS-CoV는 severe acute respiratory syndrome coronavirus 즉 심한 급성 호흡 증후군 코로나 바이러스의 약자이다.· COVD-19는 Coronavirus disease-19의 약자이고 SARS-CoV 감염으로 생긴 감염병이다. |
|
감염 연령층의 나이 |
주로 성인들이 더 잘 감염되나 소아청소년층도 감염된다. 신생아도 감염 될 수 있다 |
|
감염경로 |
SARS-CoV-2에 감염된 사람으로부터 나온 침, 콧물, 눈물 등의 비말 속에 든 SARS-CoV-2가 가까이에 있는 사람들의 코, 입 눈 등을 통해 몸속으로 들어올 때 감염된다.그 외 공기 중에 있는 SARS-CoV-2가 문고리, 전화기, 식기, 청진기, 체온기, 변소 토일렛, 키보드, 쇼핑 몰 카트 등 매개물에 묻을 수 있다. 그 묻은 SARS-CoV-2에 접촉하거나, 환자의 대변이나 소변, 침, 가래, 피에 있는 SARS-CoV가 다른사람들의 입, 코, 눈을 통해 몸속으로 들어 올때, 소변 속에 든 바이러스가 입을 통해 구강경로를 통해 몸속으로 감염될 수 있고, 피를 통해도 감염될 수 있다.임신 중 SARS-CoV-2에 감연된 산모로부터 태어난 태아나 아기에게 수직 감염되었다는 예는 아직 없다. COVD-19를 않는 산모로부터 태어난 신생아에게 모유수유를 권장하지 않는다.SARS-CoV-2에 감염된 가족이나 이 바이러스에 감염된 가족 이외 사람과 직접 간접적 또는 근접 접촉할 때 감염된 사람의 기침, 재채기, 말, 노래 등을 통해 상호 1 m 거리 이내에 있는 주위 사람들의 입, 코, 눈 등을 통해 SARS-CoV에 감염될 수 있다.이미 언급해지만, SARS-CoV-2에 오염된 청진기,체온기, 문고리, 전화기, 책상 위, 숍핑 카트 등을 만진 손으로 입, 코 눈을 만저도 간접적으로 감염될 수 있다.특히 공기가 잘 유통이 되지 않는 실내 공기 중에 있는 SARS-CoV-2 에 공기 감염 Airborne transmission이 될 수 있다.또 밀접한 실내 모임을 통해서도 감염될 수 있다. |
|
증상 징후 |
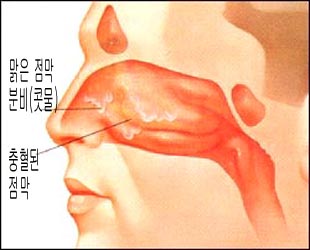
SARS-CoV-2는 주로 호흡기에 감염된다. 그래서 COVD-19가 생기고 그 감염병의 증상이 경도내지 중증도로 생길 수 있고 그러나 때로는 심하게 앓을 수도 있고 사망도 한다. 어떤 사람들은 아무 증상이 없는 무증상 COVD-19에 걸일 수 있다.냄새를 잘 맞지 못하고 음식물 맛을 잘 볼수 없는 증상도 생길 수 있다. 열, 가래가 나오지 않는 건성 기침, 피로, 코막힘, 콧물, 위장염의 증상이 생길 수 있고 더 심하면 뇌염, 심장염, 폐렴 등 많은 신체 각종 계통에 COVD-19이 생길 수 있다.성인 환자에서 볼수 있는 가슴 x 선 사진상 기관지 폐렴 영상이 나타날 수도 있다.이 병에 걸리면 자가 한정성 증상 징후가 생길 수 있다.감염된 후 3~14일간 잠복기를 거쳐 증상이 생기는 것이 보통이다.중이염에도 걸릴 수 있고 천식이 유발될 수 있다.코로나바이러스 감염병에 걸리면 콧물, 비강 울혈, 코막힘, 인두통, 재채기, 기침, 열 등의 증상 징후를 동반하는 감기의 증상과 비슷한 증상이 생길 수 있다.감기와 상기도 감염병을 일으킬 수 있는 200여종의 바이러스들 중 코로나바이러스가 가장 흔한 원인 중 하나이다.특히 영아들이나 면역 기능 손상이 있는 성인들이 사람 코로나바이러스에 감염되면 세기관지염이 생길 수도 있고 폐렴도 생길 수도 있고 그 보다 좀 더 심하게 앓을 수도 있다.중동 호흡증후군 코로나바이러스 (The Middle East Respiratory Syndrome Coronavirus /MERS-CoV)은 SARS-CoV-2 감염으로 인한 COVD-19의 일종이다.살스 감염병에 걸려 이탈리아 등 유럽 지역과 사우디아라비아 등 중동지역에서 많은 사람들에게 치명적 폐렴 환자가 발생했다. 이 바이러스에 의해서 폐렴에 걸리면 사망률이 56%나 된다고 한다.SARS-CoV-2 감염병 (중증 급성 호흡기 증후군)은 2002–2003년도에 전 세계적으로 범 유행되었고그로 인해 열, 근육통, 두통, 노근하고, 한전, 기침, 숨 가쁨 등의 증상 징후를 5~7일 동안 나타날 수 있다.설사도 하고 호흡기 부전증이 생겨 기도 내 삽입관 치료를 받아야 하는 경우도 생길 수 있다. 일 반적으로 소아 SARS-CoV-2 감염병의 증상 징후는 성인들에 비해 덜 하다.SARS-CoV-2 감염병의 일반적 사망률에 비해 60세 이상 고령자들의 경우 사망률은 상당하 높다.학동기기나 사춘기 아이들이 학교를 가지 못하고 집에 주로 가쳐 있기 때문에 우울증도 생기고 불안증이 생길 수 있다. |
|
진단 |
증상 징후, 병력, 진찰소견 등을 종합해서 이 병을 의심하면 상하 호흡기 분비물, 대변, 혈청 등에서 얻은 피검물로 종합 효소연쇄 반응 분석 Reverse transcriptase polymerase chain reaction assays(COVID-19 RT-PCR 으로 진단 할 수도 있고 SARS-CoV-2 바이러스 항원 검사로도 진단 할 수 있고SARS-CoV-2 바이러스 항체검사로 진단 할 수 있다.다. 필요에 따라 바이러스 검출해 진단한다. (www.cdc.gov/sars/index.html). 참조 |
|
치료 |
특효 약이 없다지지치료와 대증 치료를 한다.SARS-CoV-2 감염병은 Steroids, type 1 interferons, 희복기 혈청, Ribavirin, lopinavir/ritonavir 등으로 치료할 수 있으나 치료효과는 확실치 않다.만일 열이 나고 기침하고 위에 열거한 증상의 일부가 경도로 나타나면 COVID-19에 걸렸다고 추정하면서 집에 머물면서 단골의사에게 전화해 조언을 받는다. 그러면서 증상들을 적어 놓고 체온을 잰다.집안 다른 가족이나 다른 사람들에게 감염시키지 않게 주의한다.숨을 쉬기가 어렵다든지 증상이 보통이상으로 심하면 단골 의사의 지시에 따라 응급실 치료를 받는다.대부분의 경도 COVID-19 환자들은 특별한 치료를 받을 필요가 없이 자연히 회복된다. 치료를 받으러 의사를 보러 가는 이외 집을 떠나지 말고 공공장소에 가지 만다.열이 나면 약국에서 살 수 있는 타이레놀 acetaminophen등으로 집에서 자가 해열치료를 하고 육체적으로 신체적으로 쉬면서 충분한 수분을 섭취한다.필요에 따라 단골 의사에게 연락 하면서 적절한 가정 자가치료를 계속 한다. 버스나 택시 등 공중 교통 수단을 이용 하지 않는다.앓는 중 독방을 쓰고 가족들과 같은 방을 쓰지 않는다. 그리고 애완 동물과도 같이 있지 않는다.이 병을 앓기 전 그동안 근접 접촉을 했던 사람들에게 COVID-19에 걸렸다는 사실을 알린다.SARS-CoV-2에 감염되어 COVID-19 증상이 나타 나타 나기 전 2일전 부터 또는 SARS-CoV-2 진단 검사가 양성으로 나타 나기전 2일 부터 다른 사람에게 SARS-CoV-2을 감열 시킬 수 있다는 것을 알린다. 그래서 그동안 근접 접촉한 사람에게 COVID-19에 걸릴 수 있다고 알려 주어 더 이상 다른 사람에게 전파 시키는 것을 예방하고 본인도 그에 해당한 대치를 하도록 한다. 그 나라 그지방 건강 관리 당국의 지시에 따라 COVID-19를 예방치료 한다.COVID-19을 앓는 중 숨쉬기가 곤란하든지 통증이 심하든지 가슴이 아프든지, 정신이 혼동하든지 잠에서 깰 수 없든지, 얼굴이나 잎슬이 프르든지 하면 응급으로 치료를 받아야한다.필요에 따라 911전화로 도움을 받는다.다른 사람이나 애완동물과 같이 있을때는 헌겁 마스크로 코와 입을 가리고 적어도 6피트 간격을 둔다.혼자 있을 때는 마스크를 쓸 필요가 없다.2세 이하 아이들이나 숨울 잘 쉴 수 없거나 자신이 쓴 마스크를 벗을 수 없는 사람은 마스크르 쓰지 안해도 된다, 기침을 할 때는나 재채기 할 때는 티슈로 입과 코를 가린다. 그 티슈를 바로 버리고 손을 적어도 20초 동안 비누와 물로 닥거나 70% 알코올로 닥는다.
|
|
약물로 치료 한다 |
1.Remdesivir (Veklury)– 2020년 10월 22일 미 FDA 승인된 약이다. 12세 이상 병원입원 환자 혈과 주사 치료용 약. 회복 더 빠른 효과가 있다. 사망률이 적어진다. 이 약 하나로만 치료하면 충분한 치료효과가 적다.병원 입원한 COVID-19 환자를 remdesivir와 baricitinib (Olumiant) 으로 동시 치료한 결과가 더 좋다. 응급으로 치료 하는데 쓰는 약이다.Baricitinib plus Remdesivir for Hospitalized Adults with Covid-192. Dexamethasone코티코 스테로이제이다.산소호흡이나 인공호흡치료를 요하는 COVID-19환자를 경구용 또는 정맥 주사로 치료하면 사망률이 감소된다.3.Convalescent plasma2020년 8월 23일, 미 FDA의 허가를 받았다. COVID-19에서 회복되는 사람들의 회복기 플라스마(Convalescent plasma)로 치료 하면 그 속에 든 코로나 바이러스 항체로 치료된다.4. Monoclonal antibodies (MABs)체내에 들어온 바이러스와 결합해서 바이러스를 파괴 시킨다. REGN-COV2 이나 LY-CoV555 등이 있다.5. Hydroxychloroquine 과 chloroquine말라리아, 루프스, 류마토이드 병 치료에 쓰는 약이다. COVID-19치료에도 효과가 어는정도 있다.6. Azithromycin항생제의 일종이다.인플루인자, 지카 바러스 감염병 치료에도 쓰는 항생제이다. COVID-19으로 생기는 폐렴 등을 치료하는데 쓴다.7. Tocilizumab (Actemra) 와 그외 IL-6 inhibitors류마토이드 관절염이나 연소성 특발성 관절염을 치료에 쓰는 약이다.8. Interferons항 바이러스 작용이 있다9. Kinase inhibitors신체 면역 질환에 쓴다.10. Kaletra (lopinavir/ritonavir) lopinavir와 ritonavir항 바이러스 작용이 있는 약11. Ivermectin기생충 치요에 쓰는 약물이다. COVID-19 치료에도 혀과가 있다.
|
|
COVID-19를 예방, 치료 하는 Vaccine |
mRNA-1273 SARS-CoV-2 Vaccine을 개발 했고 백신 접종효과는 거의 95% 이상 효과 있다고 오늘 11/9/2020발표했다. 미국에서 아직 통상적으로 모든 사람들에게 접종하지 않고 툭수 층 사람들에게만 접종하고 있다. 접종한 후 과 즉시 알레르기 반응이 수명에게 생겼으나 치료받고 회복됐다고한다.Pfizer 회사 개발 백신 이름은 Pfizer-BioNTech COVID-19 Vaccine 이고 Moderna회사에서 만든 백신은 Moderna’s Coronavirus Vaccine이라고 한다. 그외 여러 나라에서 예방 접종을 개발 하고 있다.예방 접종의 부작용-피로. • 두통. • 근육통. • 메스꺼움. • 주사 부위의 통증. • 두통. • 피로. • 근육 및 관절 통증. • 오한. • 백신 주사를 맞은 팔의 림프절이 부어올랐습니다. • 메스꺼움과 구토. • 열. |
|
How The COVID-19 Vaccines Compare To Each OtherIn the race to end the coronavirus pandemic, the more safe, effective vaccines we have available, the sooner we’ll be able to climb out of this mess. So far, five vaccines — produced by Moderna, Pfizer, Novavax, Johnson & Johnson, and AstraZeneca — are the front-runners. In the coming months, these vaccines will not only save tons of lives, but will also alleviate some of the overwhelming pressure on the health care system and provide our frontline workers with some much-needed rest. Below, where we dissect the leading vaccines, you’ll notice the shots’ efficacy levels range from 66% to 95%. While that may seem like a huge gap, it’s worth noting that comparing vaccine efficacy levels is kind of like “comparing apples to oranges,” according to Onyema Ogbuagu, an infectious disease specialist at Yale Medicine and principal investigator of Yale’s Pfizer COVID-19 trial. Some of these vaccines (like Johnson & Johnson’s) were tested in areas where more contagious variants have taken hold, such as South Africa, whereas Moderna’s and Pfizer’s vaccines were tested before the variants identified in the U.K. and South Africa struck and began spreading like wildfire. The biggest takeaway is that while the vaccines differ in how well they prevent symptomatic disease, so far nobody who has taken any of these vaccines have died or been hospitalized due to COVID-19. Preventing severe disease is something all the shots do well, so even imperfect vaccines will prove invaluable in our quest to end the pandemic. Here’s a basic breakdown on each of the leading COVID-19 immunization options: Pfizer/BioNTechHow it works: The Pfizer shot is a messenger RNA (mRNA) vaccine, which essentially sends instructions to our cells that teach our immune system how to fight the coronavirus. Efficacy: 95% against symptomatic disease Dosage: Two shots, 21 or so days apart How well it works on COVID-19 mutations: It holds up. A lab study looking at the blood of vaccinated people found the vaccine may be slightly less effective against new variants like the one discovered in South Africa, but it still protects people well. Of course, more evidence of the shot being put to the test in the real world is necessary to validate those findings. How easily it can be tweaked to target new variants: Piece of cake. The vaccine can be changed in a couple of days, Ogbuagu said. Pfizer is already working on a booster shot against the mutations. The bigger question is what sort of testing and approval process health officials would require — that’s where there’d be delays. Side effects: Most people will feel pain and soreness in the arm where they get the shot. Up to half of people are expected to experience flu-like symptoms such as chills, fatigue and headaches, more so after the second dose. But that’s just the immune system doing its thing. How it’s stored: These mRNA vaccines are finicky, and must be stored at subzero temperatures (minus 94 degrees Fahrenheit) in freezers. Availability: The phase three trial was completed in winter 2020, and the vaccine is now being distributed to the public through emergency use authorization. Takeaway: This was the first vaccine that was approved and rolled out in United States, and the Biden administration has already purchased 100 million more doses. “At least a third of the U.S. population, roughly, will receive the dose of the Pfizer vaccine, and so definitely it will be a major contributor to curbing the epidemic here,” Ogbuagu said. ModernaHow it works: Like Pfizer, Moderna’s vaccine uses mRNA technology. Efficacy: 94.5% against symptomatic disease Dosage: Two shots, 28 or so days apart How well it works on COVID-19 mutations: The vaccine is thought to be less effective against the variant dominating in South Africa, but a lab study shows the shot can still effectively neutralize the virus and provide protection. We’ll need more data to better understand that, though. How easily it can be tweaked to target new variants: Same quick process as Pfizer. Moderna has already kicked off development and testing of a booster shot specifically targeting the variant discovered in South Africa. Side effects: Again, similar to Pfizer. Most people will feel pain and soreness in the arm where they get the shot. Up to half of people are expected to experience flu-like symptoms such as chills, fatigue and headaches, more so after the second dose. How it’s stored: These must be carefully stored at subzero temperatures (minus 4 degrees Fahrenheit) in freezers. Availability: The phase three trial was completed in winter 2020, and the vaccine is now being distributed to the public through emergency use authorization. Takeaway: The Biden administration also secured an additional 100 million doses from Moderna, so this shot will likely protect another third of the U.S. population. This puts it “on par” with the Pfizer vaccine, Ogbuagu said. NovavaxIt contains a small, harmless, synthetically produced piece of the coronavirus that trains the immune system how to recognize and dismantle the virus. It does not contain the actual live virus.Efficacy: 89.3% against symptomatic disease Dosage: Two shots, 21 days or so apart How well it works on COVID-19 mutations: Pretty good. Its efficacy is around 85.6% against the variant identified in the U.K. and 60% against the variant found in South Africa. How easily it can be tweaked to target new variants: Easily, according to experts. The Novavax team is already developing a booster dose targeting the variant found in South Africa and hopes to test it out in a few months. Side effects: Mild pain and tenderness where the shot is given. Some people may experience fatigue, headaches or muscle aches. How it’s stored: Basic refrigeration, which is plus. This makes it easier to distribute from a logistical standpoint. Availability: The phase three trial concludes in a few weeks, but it may take a couple months before the data is finalized and submitted to the Food and Drug Administration for approval. Novavax expects to be vaccinating people by May or June. Takeaway: “It will be a welcome addition to the arsenal of vaccines,” Ogbuagu said. It’s a little less efficacious compared to the mRNA vaccines, but then again, this shot was up against new variants, Ogbuagu said. Others were tested in trials before those became more dominant strains. Johnson & JohnsonHow it works: This type of shot, called a viral vector shot, uses an adenovirus (the type of virus that causes the common cold) to teach our bodies how to identify and fight the coronavirus. It does not cause you to get COVID-19. Efficacy: 66% at preventing symptomatic disease, 85% at preventing severe disease, 100% against hospitalization and death Dosage: One dose How well it works on COVID-19 mutations: Well, but overall efficacy does seem to drop with variants. The vaccine was 72% effective in U.S. trials, 66% in Latin American trials involving the variant that’s dominating Brazil and 57% in South Africa, where the B.1.351 variant has taken hold. Even so, it still provides protection against hospitalization and death. How easily it can be tweaked to target new variants: Changing viral vector vaccines isn’t *quite* as simple as modifying mRNA vaccines, but it’s still a fairly easy process without a super long timeline, according to experts. Side effects: A small percentage of people (9%) reported having a fever. Others experienced the typical symptoms: fatigue, headaches, muscle pain and injection site pain. How it’s stored: Basic refrigeration Availability: J&J plans to submit the rest of its safety and efficacy data to the FDA early this month. From there, the FDA will review the data, and if it grants emergency use authorization, people may begin receiving it later this month. Takeaway: This one-shot dose could be a game changer for vaccine access and distribution. “That’s one of the wonderful things about it, that it can get to remote and rural areas. Also, this means it can be given in doctors’ offices without having to worry about all the storage [requirements],” said Daniel Fagbuyi, an emergency physician who served as a biodefense expert in the Obama administration. Oxford/AstraZenecaHow it works: Also adenovirus-based, the AstraZeneca vaccine works like the Johnson & Johnson shot. Efficacy: 70% against symptomatic disease Dosage: Two doses. In studies, the doses were given between four to 12 weeks apart. How well it works on COVID-19 mutations: It appears to work just as well on the variant detected in the U.K., but data is still being collected on how effectively it protects against the variant identified in South Africa. How easily it can be tweaked to target new variants: Similar to Johnson & Johnson — slower than an mRNA, but still speedy. Side effects: Pain and tenderness at the injection site. Fatigue, headaches, muscle aches, chills and fever have also been reported. How it’s stored: Basic refrigeration Availability: The phase three trial was completed in fall 2020, and the vaccine is now being distributed in the European Union through emergency authorization use. It’s still being trialed in the U.S. and may not be approved for authorization from the FDA until the spring. Takeaway: The inexpensive cost of this vaccine along with its basic storage needs give AstraZeneca a big advantage. It’ll be easier for more health care providers to have the necessary supplies and get the vaccine out into the population, Fagbuyi said. Which vaccine should you get?Experts say it doesn’t really matter (and you likely won’t have a choice in most cases). Any vaccine you get should do a pretty great job at protecting you against severe illness, along with hospitalization and death. From an individual standpoint, a slight dip in efficacy might not make a big difference, but from a population standpoint, it can translate to a lot more people who remain susceptible to getting sick and spreading it to others, Ogbuagu said. All of these vaccines working together will help us achieve herd immunity. We really need 65% to 85% of the population to have protection against the virus, Ogbuagu said. And that end goal becomes a lot more realistic when we’ve got a mix of highly efficacious vaccines |
||
1/5/2921 |
Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine, NEJMA two-dose regimen of BNT162b2 conferred 95% protection against Covid-19 in persons 16 years of age or older. Safety over a median of 2 months was similar to that of other viral vaccines. (Funded by BioNTech and Pfizer; ClinicalTrials.gov number, NCT04368728. opens in a new tab.) |
|
322/2022 |
Moderna’s COVID-19 vaccine works in babies, toddlers and preschoolers, the company announced Wednesday — and if regulators agree it could mean a chance to finally start vaccinating the littlest kids by summer.
Moderna said in the coming weeks it would ask regulators in the U.S. and Europe to authorize two small-dose shots for youngsters under 6. The company also is seeking to have larger-dose shots cleared for older children and teens in the U.S. |
SIDE EFFECTS
- Fatigue.
• Headache.
• Muscle pain.
• Nausea.
• Pain at the injection site.
• Headache.
• Tiredness.
• Muscle and joint pain.
• Chills.
• Swollen lymph nodes in the arm that received the vaccine injection.
• Nausea and vomiting.
• Fever. - others
소스 및 참조 문헌
-
NEnJMed August 1, 2013, DOI: 10.1056/NEJMe1308724
-
Red Book 29th-31ㄴㅅ Ed
-
NENJ Med August 1, 2013, DOI: 0.1056/NEJMe1308724
-
JAMA 11/3/2020
-
JEJM Journal Watch 6/2020
-
Dr. Fauci: Covid-10 Vaccine for children must ‘ strike a balance’ JEJM Journal Watch 11/2020
-
AAP News Pediatrics October 22, 2020
-
Other
Copyright ⓒ 2020 John Sangwon Lee, MD., FAAP
“부모도 반의사가 되어야 한다”-내용은 여러분들의 의사로부터 얻은 정보와 진료를 대신할 수 없습니다.
“The information contained in this publication should not be used as a substitute for the medical care and advice of your doctor. There may be variations in treatment that your doctor may recommend based on individual facts and circumstances.
“Parental education is the best medicine.
2. 이전 정보
원인
사람 코로나바이러스(Human coronaviruses/HCoVs)는
HCoVs 229E,
HCoVs OC43,
HCoVsNL63,
HCoVs HKU1 등 여러 종으로 분리된다.
이중 한 가지 사람 코로나바이러스에 감염되면 사람 코로나바이러스 감염병에 걸릴 수 있다. 이 감염병은 주로 겨울철과 봄철에 유행한다.
그 중 살스–코로나바이러스(SARS-CoV)에 감염되면 살스 감염병(중증 급성 호흡기 증후군)에 걸릴 수 있다.
박쥐나 고양이 등 중간 숙주에 있었던 SARS-CoV 유사 바이러스가 새로 진화되어 새로운 바이러스가 생겼다고 믿고 있다.
증상 징후
이 병에 걸리면 자가 한정성 증상 징후가 생길 수 있다.
3~4일 간 앓는 것이 보통이다. 중이염에도 걸릴 수 있고 천식이 유발될 수 있다.
좀 더 심한 사람 코로나바이러스 감염병에 걸리면 콧물, 비강 울혈, 코막힘, 인두통, 재채기, 기침, 열 등의 증상 징후를 동반하는 감기와 상기도 감염병이 생길 수 있다.
사실은 감기와 상기도 감염병을 일으킬 수 있는 200여종의 바이러스들 중 사람 코로나바이러스가 가장 흔한 원인이다.
특히 영아들이나 면역 기능 손상이 있는 사람들이나 또는 성인들이 사람 코로나바이러스에 감염되면 세기관지염이 생길 수도 있고 폐렴에 걸릴 수도 있다. 그 보다 좀 더 심하게 앓을 수도 있다.
살스 감염병(중증 급성 호흡기 증후군/Sars)에 걸려 사망할 수도 있다.
중동 호흡증후군 코로나바이러스(The Middle East respiratory syndrome coronavirus /MERS-CoV)는 주로 사람에서 사람으로 전파된다고 믿는다. 살스 감염병에 걸려 이탈리아 등 유럽 지역과 사우디아라비아 등 중동지역에서 여러 명의 치명적 폐렴 환자가 발생했다. 이 바이러스에 의해서 폐렴에 걸리면 사망률이 56%나 된다고 한다. 소스: N Engl J Med August 1, 2013 DOI: 10.1056/NEJMe1308724
SARS-CoV 감염병(중증 급성 호흡기 증후군)은 2002–2003년도에 전 세계적으로 범 유행되었고 그로 인해 고열, 근육통, 두통, 노근하고, 한전, 기침, 숨 가쁨 등의 증상 징후를 5~7일 동안 나타날 수 있다. 그리고 설사도 하고 호흡기 부전증이 생겨 기도 내 삽입관 치료를 받아야 했다.
소아 SARS-CoV 감염병의 증상 징후는 덜 하다.
SARS-CoV 감염병의 일반적 사망률은 10%이나 60세 이상 고령자들의 경우 사망률은 거의 50%이었다.
진단
증상 징후, 병력, 진찰소견 등을 종합해서 이 병을 의심하면 상하 호흡기 분비물, 대변, 혈청 등 피검물로 중합 효소연쇄 반응 분석해 진단 할 수도 있다.
필요에 따라 바이러스 검출로 확진한다, comprehensive respiratory molecular diagnostic testing for non-SARS HCoVs using reverse transcriptase polymerase chain reaction assays으로 진단 할 수 있다.(www.cdc.gov/sars/index.html). 참조
치료
항 바이러스제나 스테로이드 치료에 치료되지 않는다. 지지치료와 대증 치료를 한다.
ARS-CoV 감염병은 Steroids, type 1 interferons, 희복기 혈청, ribavirin, lopinavir/ritonavir 등으로 치료할 수 있으나 치료효과는 확실치 않다.
코로나바이러스 (SARS-Severe acute respiratory syndrome -Covid-2) 감염병 Covid-19
1. Latest information Coronavirus (SARS-Severe acute respiratory syndrome -Covid-2) Infectious disease Covid-19 
corona virus video Source: US CDC
• If you suspect this disease by summarizing symptom signs, medical history, and examination findings, a comprehensive enzyme chain reaction analysis with specimens obtained from upper and lower respiratory secretions, feces, serum, etc. Reverse transcriptase polymerase chain reaction assays (COVID-19 RT-PCR) It can also be diagnosed with SARS-CoV-2 virus antigen test. • It can be diagnosed with SARS-CoV-2 virus antibody test. • All. Virus detection and diagnosis as necessary. (www.cdc.gov/sars/index.html). Reference
There are seven species of human coronaviruses (HCoVs).
There are seven species of human coronaviruses (HCoVs). |
1. HCoV-229E,2. HCoV-OC43,3. HCoV-NL63,4. HCoV-HKU1,5. severe acute respiratory syndrome coronavirus (SARS-CoV),6. Middle East respiratory syndrome coronavirus (MERS-CoV)7. Novel coronavirus (2019-nCoV, a.k.a. SARS-CoV-2). |
|
SARS-CoV-2 and COVD-19 are abbreviations. |
SARS-CoV is an abbreviation for severe acute respiratory syndrome coronavirus. · COVD-19 is an abbreviation for Coronavirus disease-19 and is an infectious disease caused by SARS-CoV infection. |
|
age of infection |
Adults are more likely to be infected, but children and adolescents are also infected. Newborns can also be infected |
|
epidemiology |
• SARS-CoV-2 in droplets from a person infected with SARS-CoV-2, such as saliva, runny nose, and tears, enters the body through the nose, mouth, or eyes of people nearby.• In addition, SARS-CoV-2 in the air may be deposited on objects such as doorknobs, telephones, tableware, stethoscopes, thermometers, toilet toilets, keyboards, and shopping mall carts. When SARS-CoV-2 is contaminated, or SARS-CoV in a patient’s feces, urine, saliva, sputum, or blood enters the body through the mouth, nose, or eyes of others, the virus in the urine is transmitted through the mouth. It can be transmitted into the body through the oral route and can also be transmitted through blood.• There are no cases of vertical transmission of fetuses or babies born to mothers infected with SARS-CoV-2 during pregnancy. Breastfeeding is not recommended for newborns born to mothers who do not have COVID-19.• When in direct, indirect or close contact with a family member infected with SARS-CoV-2 or with someone other than a family member infected with this virus, the mouth and nose of those around you within 1 m of each other through the infected person’s cough, sneeze, speech or song. SARS-CoV can be transmitted through, eye, etc.• As already mentioned, if you touch your mouth, nose, or eyes with your hands, such as a stethoscope, thermostat, doorknob, phone, desk, or shopping cart contaminated with SARS-CoV-2, you can indirectly get infected.• Airborne transmission of SARS-CoV-2 is possible, especially in indoor air with poor air circulation.• It can also be transmitted through close indoor gatherings. |
|
Symptoms and signs |
• SARS-CoV-2 mainly infects the respiratory system. So, COVD-19 occurs and the symptoms of the infectious disease can be mild or severe, but sometimes you can get seriously ill and even die. Some people can get asymptomatic COVD-19 asymptomatic.
• You may also experience symptoms such as not being able to smell well and not being able to taste food well. Symptoms of fever, dry cough without sputum, fatigue, stuffy nose, runny nose, and gastroenteritis can occur, and even worse, COVD-19 can occur in many systems of the body, such as encephalitis, heart disease, and pneumonia. • A chest x-ray seen in adult patients may show images of bronchopneumonia. • People with this disease may develop self-limiting symptoms. • Symptoms usually develop after an incubation period of 3 to 14 days after infection. • You can also get otitis media and cause asthma. • A coronavirus infection can cause symptoms similar to those of the common cold, with symptoms such as runny nose, nasal congestion, stuffy nose, sore throat, sneezing, coughing and fever. • Of the more than 200 viruses that can cause colds and upper respiratory tract infections, coronavirus is one of the most common causes. • Bronchiolitis, pneumonia, or even worse, can develop when infected with human coronavirus, especially in infants and adults with compromised immune function. • The Middle East Respiratory Syndrome Coronavirus (MERS-CoV) is a type of COVD-19 caused by SARS-CoV-2 infection. • As a result of the SARS infection, many people in Europe, including Italy, and the Middle East, including Saudi Arabia, suffered fatal pneumonia. Pneumonia caused by this virus has a 56% mortality rate. • SARS-CoV-2 infectious disease (severe acute respiratory syndrome) was prevalent worldwide in 2002–2003 • As a result, symptoms such as fever, muscle pain, headache, tiredness, shivering, cough, and shortness of breath may appear for 5 to 7 days. • Diarrhea and respiratory failure may occur, requiring treatment with an endotracheal tube. In general, the symptoms of SARS-CoV-2 infection in children are less than in adults. • Compared to the general mortality rate from SARS-CoV-2 infectious disease, the mortality rate for the elderly over 60 is significantly higher. • Depression and anxiety can also occur because schoolchildren and adolescents are often at home without going to school. |
|
Diagnosis |
• If you suspect this disease by summarizing symptom signs, medical history, and examination findings, a comprehensive enzyme chain reaction analysis with specimens obtained from upper and lower respiratory secretions, feces, serum, etc. Reverse transcriptase polymerase chain reaction assays (COVID-19 RT-PCR) It can also be diagnosed with SARS-CoV-2 virus antigen test.• It can be diagnosed with SARS-CoV-2 virus antibody test.• All. Virus detection and diagnosis as necessary. (www.cdc.gov/sars/index.html). Reference |
|
Treatment |
|
|
Drug treatment |
1.Remdesivir (Veklury) – FDA-approved drug on October 22, 2020. Medicines for blood and injection treatment of hospitalized patients 12 years of age or older. Recovery is faster. the mortality rate is reduced. Treatment with this drug alone is insufficiently effective. Simultaneous treatment of hospitalized COVID-19 patients with remdesivir and baricitinib (Olumiant) has better results. It is a drug used for emergency treatment. Baricitinib plus Remdesivir for Hospitalized Adults with Covid-192. Dexamethasone It is a corticosteroid. Oral or intravenous treatment of COVID-19 patients requiring oxygen respiration or artificial respiration reduces mortality.3.Convalescent plasma On August 23, 2020, it was approved by the US FDA. When people recovering from COVID-19 are treated with convalescent plasma, they are treated with the coronavirus antibodies contained therein.4. Monoclonal antibodies (MABs) It binds to the virus that enters the body and destroys the virus. REGN-COV2 or LY-CoV555.5. Hydroxychloroquine and chloroquine It is used to treat malaria, lupus, and rheumatoid diseases. It is also somewhat effective in treating COVID-19.6. Azithromycin It is a type of antibiotic. It is an antibiotic used to treat influenza and Zika virus infections. It is used to treat pneumonia caused by COVID-19.7. Tocilizumab (Actemra) and other IL-6 inhibitors It is a drug used to treat rheumatoid arthritis or juvenile idiopathic arthritis.8. Interferons have antiviral action9. Kinase inhibitors It is used for body immune diseases.10. Kaletra (lopinavir/ritonavir) lopinavir and ritonavir drugs with antiviral action 11. Ivermectin It is a drug used to treat parasites. There is a linguistic department in the treatment of COVID-19.
|
|
COVID-19 Vaccine |
Vaccinations Vaccines in the U.S. Developed mRNA-1273 SARS-CoV-2 Vaccine, and announced today 11/9/2020 that the vaccination effect is almost 95% effective. In the United States, it is still not routinely vaccinated to all people, but only to the upper class. It is said that an allergic reaction occurred in the lifespan immediately after vaccination, but it was treated and recovered. The vaccine developed by the Pfizer company is named Pfizer-BioNTech COVID-19 Vaccine. The vaccine made by Moderna is called Moderna’s Coronavirus Vaccine. Vaccinations are being developed in several other countries. |
|
How The COVID-19 Vaccines Compare To Each OtherIn the race to end the coronavirus pandemic, the more safe, effective vaccines we have available, the sooner we’ll be able to climb out of this mess. So far, five vaccines — produced by Moderna, Pfizer, Novavax, Johnson & Johnson and AstraZeneca — are the front-runners. In the coming months, these vaccines will not only save tons of lives, but they will also alleviate some of the overwhelming pressure on the health care system and provide our frontline workers with some much-needed rest. Below, where we dissect the leading vaccines, you’ll notice the shots’ efficacy levels range from 66% to 95%. While that may seem like a huge gap, it’s worth noting that comparing vaccine efficacy levels is kind of like “comparing apples to oranges,” according to Onyema Ogbuagu, an infectious disease specialist at Yale Medicine and principal investigator of Yale’s Pfizer COVID-19 trial. Some of these vaccines (like Johnson & Johnson’s) were tested in areas where more contagious variants have taken hold, such as South Africa, whereas Moderna’s and Pfizer’s vaccines were tested before the variants identified in the U.K. and South Africa struck and began spreading like wildfire. The biggest takeaway is that while the vaccines differ in how well they prevent symptomatic disease, so far nobody who has taken any of these vaccines have died or been hospitalized due to COVID-19. Preventing severe disease is something all the shots do well, so even imperfect vaccines will prove invaluable in our quest to end the pandemic. Here’s a basic breakdown on each of the leading COVID-19 immunization options: Pfizer/BioNTech How it works: The Pfizer shot is a messenger RNA (mRNA) vaccine, which essentially sends instructions to our cells that teach our immune system how to fight the coronavirus. Efficacy: 95% against symptomatic disease Dosage: Two shots, 21 or so days apart How well it works on COVID-19 mutations: It holds up. A lab study looking at the blood of vaccinated people found the vaccine may be slightly less effective against new variants like the one discovered in South Africa, but it still protects people well. Of course, more evidence of the shot being put to the test in the real world is necessary to validate those findings. How easily it can be tweaked to target new variants: Piece of cake. The vaccine can be changed in a couple of days, Ogbuagu said. Pfizer is already working on a booster shot against the mutations. The bigger question is what sort of testing and approval process health officials would require — that’s where there’d be delays. Side effects: Most people will feel pain and soreness in the arm where they get the shot. Up to half of people are expected to experience flu-like symptoms such as chills, fatigue and headaches, more so after the second dose. But that’s just the immune system doing its thing. How it’s stored: These mRNA vaccines are finicky, and must be stored at subzero temperatures (minus 94 degrees Fahrenheit) in freezers. Availability: The phase three trial was completed in winter 2020, and the vaccine is now being distributed to the public through emergency use authorization. Takeaway: This was the first vaccine that was approved and rolled out in United States, and the Biden administration has already purchased 100 million more doses. “At least a third of the U.S. population, roughly, will receive the dose of the Pfizer vaccine, and so definitely it will be a major contributor to curbing the epidemic here,” Ogbuagu said. Moderna How it works: Like Pfizer, Moderna’s vaccine uses mRNA technology. Efficacy: 94.5% against symptomatic disease Dosage: Two shots, 28 or so days apart How well it works on COVID-19 mutations: The vaccine is thought to be less effective against the variant dominating in South Africa, but a lab study shows the shot can still effectively neutralize the virus and provide protection. We’ll need more data to better understand that, though. How easily it can be tweaked to target new variants: Same quick process as Pfizer. Moderna has already kicked off development and testing of a booster shot specifically targeting the variant discovered in South Africa. Side effects: Again, similar to Pfizer. Most people will feel pain and soreness in the arm where they get the shot. Up to half of people are expected to experience flu-like symptoms such as chills, fatigue and headaches, more so after the second dose. How it’s stored: These must be carefully stored at subzero temperatures (minus 4 degrees Fahrenheit) in freezers. Availability: The phase three trial was completed in winter 2020, and the vaccine is now being distributed to the public through emergency use authorization. Takeaway: The Biden administration also secured an additional 100 million doses from Moderna, so this shot will likely protect another third of the U.S. population. This puts it “on par” with the Pfizer vaccine, Ogbuagu said. Novavax How it works: Novavax’s shot is a protein subunit vaccine. It contains a small, harmless, synthetically produced piece of the coronavirus that trains the immune system how to recognize and dismantle the virus. It does not contain the actual live virus. Efficacy: 89.3% against symptomatic disease Dosage: Two shots, 21 days or so apart How well it works on COVID-19 mutations: Pretty good. Its efficacy is around 85.6% against the variant identified in the U.K. and 60% against the variant found in South Africa. How easily it can be tweaked to target new variants: Easily, according to experts. The Novavax team is already developing a booster dose targeting the variant found in South Africa and hopes to test it out in a few months. Side effects: Mild pain and tenderness where the shot is given. Some people may experience fatigue, headaches or muscle aches. How it’s stored: Basic refrigeration, which is plus. This makes it easier to distribute from a logistical standpoint. Availability: The phase three trial concludes in a few weeks, but it may take a couple months before the data is finalized and submitted to the Food and Drug Administration for approval. Novavax expects to be vaccinating people by May or June. Takeaway: “It will be a welcome addition to the arsenal of vaccines,” Ogbuagu said. It’s a little less efficacious compared to the mRNA vaccines, but then again, this shot was up against new variants, Ogbuagu said. Others were tested in trials before those became more dominant strains. Johnson & Johnson How it works: This type of shot, called a viral vector shot, uses an adenovirus (the type of virus that causes the common cold) to teach our bodies how to identify and fight the coronavirus. It does not cause you to get COVID-19. Efficacy: 66% at preventing symptomatic disease, 85% at preventing severe disease, 100% against hospitalization and death Dosage: One dose How well it works on COVID-19 mutations: Well, but overall efficacy does seem to drop with variants. The vaccine was 72% effective in U.S. trials, 66% in Latin American trials involving the variant that’s dominating Brazil and 57% in South Africa, where the B.1.351 variant has taken hold. Even so, it still provides protection against hospitalization and death. How easily it can be tweaked to target new variants: Changing viral vector vaccines isn’t *quite* as simple as modifying mRNA vaccines, but it’s still a fairly easy process without a super long timeline, according to experts. Side effects: A small percentage of people (9%) reported having a fever. Others experienced the typical symptoms: fatigue, headaches, muscle pain and injection site pain. How it’s stored: Basic refrigeration Availability: J&J plans to submit the rest of its safety and efficacy data to the FDA early this month. From there, the FDA will review the data, and if it grants emergency use authorization, people may begin receiving it later this month. Takeaway: This one-shot dose could be a game changer for vaccine access and distribution. “That’s one of the wonderful things about it, that it can get to remote and rural areas. Also, this means it can be given in doctors’ offices without having to worry about all the storage [requirements],” said Daniel Fagbuyi, an emergency physician who served as a biodefense expert in the Obama administration. Oxford/AstraZeneca How it works: Also adenovirus-based, the AstraZeneca vaccine works like the Johnson & Johnson shot. Efficacy: 70% against symptomatic disease Dosage: Two doses. In studies, the doses were given between four to 12 weeks apart. How well it works on COVID-19 mutations: It appears to work just as well on the variant detected in the U.K., but data is still being collected on how effectively it protects against the variant identified in South Africa. How easily it can be tweaked to target new variants: Similar to Johnson & Johnson — slower than an mRNA, but still speedy. Side effects: Pain and tenderness at the injection site. Fatigue, headaches, muscle aches, chills and fever have also been reported. How it’s stored: Basic refrigeration Availability: The phase three trial was completed in fall 2020, and the vaccine is now being distributed in the European Union through emergency authorization use. It’s still being trialed in the U.S. and may not be approved for authorization from the FDA until the spring. Takeaway: The inexpensive cost of this vaccine along with its basic storage needs give AstraZeneca a big advantage. It’ll be easier for more health care providers to have the necessary supplies and get the vaccine out into the population, Fagbuyi said. Which vaccine should you get?Experts say it doesn’t really matter (and you likely won’t have a choice in most cases). Any vaccine you get should do a pretty great job at protecting you against severe illness, along with hospitalization and death. From an individual standpoint, a slight dip in efficacy might not make a big difference, but from a population standpoint, it can translate to a lot more people who remain susceptible to getting sick and spreading it to others, Ogbuagu said. All of these vaccines working together will help us achieve herd immunity. We really need 65% to 85% of the population to have protection against the virus, Ogbuagu said. And that end goal becomes a lot more realistic when we’ve got a mix of highly efficacious vaccines |
||
1/5/2021 |
Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine, NEJMA two-dose regimen of BNT162b2 conferred 95% protection against Covid-19 in persons 16 years of age or older. Safety over a median of 2 months was similar to that of other viral vaccines. (Funded by BioNTech and Pfizer; ClinicalTrials.gov number, NCT04368728. opens in new tab.) |
|
3/29/2022 Update
U.S. regulators on Tuesday authorized another COVID-19 booster for people age 50 and older, a step to offer extra protection for the most vulnerable in case the coronavirus rebounds.
The Food and Drug Administration’s decision opens a fourth dose of the Pfizer or Moderna vaccines to those people at least four months after their previous booster.
Until now, the FDA had cleared fourth doses only for people 12 and older who have severely weakened immune systems. The agency said this especially fragile group also can get an additional booster, a fifth shot.
Update 5./19/2022
CDC advisers recommend booster shots for kids 5 to 11
Kids a few years out of booster seats may soon be getting booster shots.
A Centers for Disease Control and Prevention advisory panel voted Thursday to recommend that children ages 5 to 11 receive Pfizer booster shots, four months after the agency endorsed extra jabs for Americans over 12.
The CDC typically follows the recommendations of its advisory committees. Dr. Rochelle Walensky, the CDC director, is expected to promptly follow the latest guidance.
The Advisory Committee on Immunization Practices voted 11 to 2 in favor of children over 5 receiving a booster shot at least five months after their initial inoculation series. One member of the panel abstained.
The vote came at the close of a five-hour virtual meeting. “If and when CDC concurs with this recommendation we will send a notice out,” Kristen Nordlund, a CDC spokeswoman, said in an email following the vote.
Booster shots provide rugged protection against the meddlesome coronavirus mutations that have continued to move through the population in the pandemic’s third year.
New York is currently riding out a wave of the virus powered by fast-spreading subvariants of the omicron strain, and officials have urged complete vaccination.
“The best way to stay out of the hospital with COVID-19 is by keeping up to date with your vaccination and booster doses,” Gov. Hochul said in a statement Thursday, as her office announced 19 more coronavirus deaths.
The state’s weeklong test positivity sat at a striking 7.7% on Thursday, according to government figures, up from 1.9% two months ago. In New York City, the rate was slightly lower — 5.4% — but rising, according to the governor’s office.
About 39% of all New Yorkers have received booster shots, according to state data. And only about 37% of New York children ages 5 to 11 have completed their initial vaccination series, according to the state Health Department.
Dr. Denis Nash, a professor of epidemiology at the City University of New York, said the CDC panel’s recommendation marked a “really important development” given the state’s recent spike in cases. He encouraged parents to take advantage.
“There’s no really no real downside,” Nash said, “and only upside of protecting kids against severe disease and in some rare cases death.”
UPDATE 6/17/2022 Yahoo News
FDA authorizes 1st COVID-19 shots for infants, preschoolers
U.S. regulators on Friday authorized the first COVID-19 shots for infants and preschoolers, paving the way for vaccinations to begin next week.
The Food and Drug Administration’s action follows its advisory panel’s unanimous recommendation for the shots from Moderna and Pfizer. That means U.S. kids under 5 — roughly 18 million youngsters — are eligible for the shots, about 1 1/2 years after the vaccines first became available in the U.S. for adults, who have been hit the hardest during the pandemic.
There’s one step left: The Centers for Disease Control and Prevention recommends how to use vaccines and its vaccine advisers are set to discuss the shots Friday and vote on Saturday. A final signoff would come from CDC Director Dr. Rochelle Walensky.
At a Senate hearing Thursday, Walensky said her staff was working over the Juneteenth federal holiday weekend “because we understand the urgency of this for American parents.”
She said pediatric deaths from COVID-19 have been higher than what is generally seen from the flu each year.
“So I actually think we need to protect young children, as well as protect everyone with the vaccine and especially protect elders,” she said.
The FDA also authorized Moderna’s vaccine for school-aged children and teens. Pfizer’s shots had been the only option for those ages.
For weeks, the Biden administration has been preparing to roll out the vaccines for little kids, with states, tribes, community health centers and pharmacies allowed to preorder millions of doses. FDA’s emergency use authorization allows manufacturers to begin shipping vaccine across the country. Vaccinations could begin early next week.
While young children generally don’t get as sick from COVID-19 as older kids and adults, their hospitalizations surged during the omicron wave and FDA’s advisers determined that benefits from vaccination outweighed the minimal risks. Studies from Moderna and Pfizer showed side effects, including fever and fatigue, were mostly minor.
“As we have seen with older age groups, we expect that the vaccines for younger children will provide protection from the most severe outcomes of COVID-19, such as hospitalization and death,” FDA Commissioner Robert Califf said in a statement.
The two vaccine brands use the same technology but there are differences.
Pfizer’s vaccine for kids younger than 5 is one-tenth of the adult dose. Three shots are needed: the first two given three weeks apart and the last at least two months later.
Moderna’s is two shots, each a quarter of its adult dose, given about four weeks apart for kids under 6.
The vaccines are for children as young as 6 months. Moderna next plans to study its shots for babies as young as 3-months-old. Pfizer has not finalized plans for shots in younger infants. A dozen countries, including China, already vaccinate kids under 5.
Dr. Beth Ebel, professor of pediatrics at University of Washington in Seattle, said the tot-sized vaccines would be especially welcomed by U.S. parents with children in daycare where outbreaks can sideline parents from jobs, adding to financial strain.
“A lot of people are going to be happy and a lot of grandparents are going to be happy, too, because we’ve missed those babies who grew up when you weren’t able to see them,” Ebel said.
___
AP Medical Writers Laura Ungar and Carla K. Johnson contributed.
___
Follow AP Medical Writer Lindsey Tanner at @LindseyTanner.
___
The Associated Press Health and Science Department receives support from the Howard Hughes Medical Institute’s Department of Science Education. The AP is solely responsible for all content.
출처 및 참조 문헌 Sources and references
- NelsonTextbook of Pediatrics 22ND Ed
- The Harriet Lane Handbook 22ND Ed
- Growth and development of the children
- Red Book 32nd Ed 2021-2024
- Neonatal Resuscitation, American Academy Pediatrics
-
N Engl J Med August 1, 2013, DOI: 10.1056/NEJMe1308724
-
Red Book 29th Ed
-
Others
- Copyright ⓒ 2014 John Sangwon Lee, MD., FAAP
- “부모도 반의사가 되어야 한다”-내용은 여러분들의 의사로부터 얻은 정보와 진료를 대신할 수 없습니다.
- “The information contained in this publication should not be used as a substitute for the medical care and advice of your doctor. There may be variations in treatment that your doctor may recommend based on individual facts and circumstances.
- “Parental education is the best medicine.